AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Reactivity trend in alkaline earth1/18/2024 
This is because metal hydroxides thermally decompose to give the metal oxide and water. When metal reacts with cold water, we get metal hydroxide and when metal reacts with steam, the metal oxide is formed. So, it reacts vigorously with water at room temperature. It requires the least ionisation energy and hence activation energy to form ions. Barium (Ba) is the second last alkaline earth metal and is the heaviest metal in its group. Consequently, this leads to lower activation energy and therefore, faster reactions. Barium forms barium peroxide (BaO 2 ) because the larger O 2 2 ion is better able to separate the large Ba 2+ ions in the crystal lattice. All group 2 elements except barium react directly with oxygen to form the simple oxide MO. Thus, the reaction becomes easier as the energy needed to form positive ions falls. The reactions of the alkaline earth metals with oxygen are less complex than those of the alkali metals. First and second ionisation energies are required to convert the metal atoms into ions with a +2 charge.Īs we go down the group, ionisation energy decreases because it gets easier to form the ions due to less attraction of electrons towards the nucleus in heavier atoms. The formation of these ions from the original metal requires input of energy. When alkaline earth metals react with water to form metal oxides or hydroxides, metal ions are formed. Alkaline earth metals become more reactive towards water as we go down the group. Magnesium and beryllium react with steam to give metal oxide and hydrogen gas. Group 2 elements react with cold water, except beryllium and magnesium, to give metal hydroxide and water. As you go down group 1 (the alkali metals) in the periodic table, the elements get more reactive. It can be used as a differentiated activity for the more able students within a group. 
This could be used to follow up some work on the periodic table where the trends in reactivity in groups 1 and 7 have been identified. Group 2 elements comprise beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr), and barium (Ba) and these elements are known as alkaline earth metals. Reactivity of halogens and alkali metals. Chemistry for the gifted and talented: trends in reactivity in the periodic table. As we go down the group, group 2 elements become more reactive towards water. There is a trend in the reactivity of alkaline earth metals with water. Recall the elements that constitute group 2. 
Hint: Group 2 elements are called alkaline earth metals.
0 Comments
Read More
Back to Blog
Robert kiyosaki cashflow game pc1/18/2024 
Unfortunately, what they’re missing (or in some cases trying to block) is that the power of play is exactly what unlocks the ability for anyone to achieve their fullest potential. They want you to work, work, work as hard as you can. 
And, as we can see from the example of those play labs in Bangladesh, those people often do not want you to play or have fun. In the Rat Race, you rely on other people to mind your business for you. And it completely shuts off your mind from other possibilities and ways of living and thriving in the world. The reason for this is because the Rat Race makes you entirely dependent on other people: your parents, your teachers, your boss, etc. Unfortunately, the Rat Race game is a losing one. In short, it’s the vicious cycle we learn from a young age that the only way to get ahead is to go to a good school, get a good job, and work harder and harder to try and get ahead. In his book, Rich Dad Poor Dad, Robert writes about the concept of the Rat Race. What would lead so many adults to be so afraid of play? 
Unsurprisingly, these aren’t uncommon comments that may be heard when preaching the importance of play as the most important way to learn, better than books, lectures, and tests. “I’d rather do housework than waste my time with play…” ‘What’s the point of this’ and ‘What’s the use of play?’ were common refrains…” “There’s a preconception that from the early years are led into a world of textbooks, and there’s no room for them to play.” In an article published by Quartz on the importance of play in learning and an experiment with play labs in Bangladesh, you can find the following quotes: Sadly, this is still the way many educators and parents across the world respond to the idea of children playing in order to learn. They have more important subjects to learn.” Clearly, they didn’t understand the power of play. Their verbal reply was, “We do not play games in school, and we are not interested in teaching young people about money. In 1996, Robert Kiyosaki’s educational board game, CASHFLOW®, was submitted to a group of instructors at a prominent university for their feedback. The best way to learn how to get out of the Rat Race is by playing the educational board game, CASHFLOW® There have been proven results in the power of play when it comes to learning Traditional methods of teaching go against the natural way in which humans are meant to learn
Back to Blog
Color grabber laundry detergent1/18/2024 
I would love to hear your opinions about it too, so if you've tried one of them you can skip down to share your review here. Here's some reviews of these products which address these very issues. 200 Sheets Laundry Detergent,Savings for Wash,Keep Clothes Original Piece Color Catch Collector Keeper Stain Removing Grabber Maintains Homecoming Familie Dress Easy to Carry and Use,Cruelty Free Safe Material: Non-Woven Quantity: 200 Pieces Single Piece Size: 10 x 14 cm / 3.94 x 5.51 inches Weight: 5. It is obviously more frugal just to sort your clothes into various piles instead of using this product. In addition, these products aren't cheap. What I'm still trying to decide is if they collect enough dye to make it worth my while to use them, or if I'm just slowly making my clothes dingy by not sorting. Using color grabber sheets for your laundry is particularly important when washing new, vibrant, or dark-colored garments that have a higher chance of bleeding. You can see that, for example, from the dark color of the sheet after the wash (see picture above). Shout Color Catcher sheets absorb and trap loose dyes in your wash water, protecting clothes against color bleeds and helping preserve original colors. I've personally tried some of these products (see my reviews below, along with other people's) and I have seen that they do collect dye. Here's the video from Consumer Reports showing their tests of two of the most popular brands of this product, Shout Color Catcher and Carbona Color & Dirt Grabber, and how they worked in comparison to just washing the clothes without the product included. While some people swear by these cloths (see some of the reviews below, for example) other authorities (namely Consumer Reports) say that these products just don't work as promised, and you should just save your money and sort your laundry like we've always been told to do before. Colour Catcher is a revolutionary laundry sheet with a. The short answer to this is that people disagree. Coloured laundry releases dye particles in the water during the wash and bleed into your laundry. The million dollar question, however, is do they really work as promised? Use 2 sheets for large loads or loads with new, dark, or non-colorfast garments. I have also purchased two 5 liters can of liquid laundry detergent. Place 1 sheet in washing machine then add detergent and clothing. These color catchers promise a lot, like being able to wash all your clothes together, including a bright red garment with your whites. The product is sold with good discount of 70 which is affordable for any middleclass family. The cloths don't do that, but instead are just supposed to grab dye from the wash water. Please note that these catchers are not the same as dye or color remover products, which grab dye from fabric itself. The Velcro® closures easily open and close-meaning no more hassles with finicky zips.Therefore, a special type of laundry product has been created to try to fix this problem - little pieces of cloth specially formulated to grab bleeding dyes in the wash water and keep them from redepositing on other clothes. 
We source the finest ingredients & adjust our formulas to ensure the best laundry detergent. Powerful powder pods tackle your most difficult laundry challenges, without harmful toxins or additives. If using Quilt Wash, mix two scoops per gallon of lukewarm water and stir until it is. Resist the urge to use more detergent than necessary, which will require extensive rinsing to remove and can leave residue behind that will lend a dingy, dull appearance to the quilt. Package design clearly states what the product does & how to use it the ingredients are listed in full, along with each ingredient's purpose. liquid laundry detergent, depending on the size of the quilt. Just like all of our products, Grab Green Pods are filled with non-toxic ingredients, making them the safest choice for your household. And not only do they not look like candy, but the pod’s extremely bitter outer film will keep anyone from wanting to take a bite. While liquid pods are colorful and squishy, ours are intentionally white and made of powder so they don’t look like irresistibly colorful candy and are much more difficult to ingest. 1299 (0.16/Count) FREE delivery Tue, Nov 21 on 35 of items shipped by Amazon. 
Unlike conventional liquid pods that come with too many risks, Grab Green Pods have four layers of security in place to protect you and your loved ones. Color Catcher Laundry Sheets by Zero Trace - 80 Count - Prevents Dye Transfer & Keeps Clothes Original Color - Fragrance-Free, Plant-Based - Ideal for Mixed Washes. Our powder pods were designed with your ultimate safety in mind.
Back to Blog
Straight flow hair1/18/2024 There are some differences in bottled sea salt sprays compared to ocean water.įor starters, the salt in ocean water is heavily composed of sodium chloride-what we know as table salt. And we wouldn’t recommend going to the ocean and filling up a spray bottle with the intention of using it in your hair. There’s a little more to sea salt sprays than just mixing salt and water. What Are The Benefits of Using Sea Salt Spray? This can allow you to go longer between washing with shampoo, which can actually help your hair retain more natural moisture.Īloe, in particular, is known for its anti-inflammatory properties, and many Beardbrand Sea Salt Spray users report that it has helped them prevent and control dandruff.īeardbrand Sea Salt Spray adds texture and enhances the natural waviness of hair. Sea Salt Spray helps remove toxins from your scalp and hairĮach of the key ingredients in Beardbrand Sea Salt Spray-magnesium sulfate, magnesium chloride, aloe, and kaolinite-help to remove toxins, dirt, and debris from the scalp and hair. Sea salt sprays will add volume to straight hair, but they won’t help curls magically appear. Type 1 hair is generally resistant to curling without some chemical assistance. Unfortunately, if you have type 1 hair, you won’t get much curl from using a sea salt spray. This curl-enhancing effect tends to work best for those with types 2, 3, and 4.Īpply Sea Salt Spray to wet hair and scrunched with the hand, and the hair shafts will settle deeper into their curl pattern as they dry. This helps the hair’s natural curl stay in place a little tighter. In Beardbrand Sea Salt Spray, the ingredients magnesium chloride, magnesium sulfate, and kaolinite adhere to the hair strands and apply a small amount of hold. Type 1 has no curl, type 2 has a natural “s” shape, type 3 has round ringlets, and type 4 is more of a "z” shape with sharp angles. In the simplest form, type 1 hair is straight, type 2 hair is wavy, type 3 hair is curly, and type 4 hair is coily.Įveryone’s hair has a natural curl pattern. Hair types are often characterized by their density, texture, and curl pattern. Sea Salt Spray enhances the natural curl pattern of your hair The salt, which naturally has a slight amount of grit, helps texturize the hair. Naturally, this allows for greater volume because the hair shafts aren’t weighed down by the oil. Sea Salt Spray helps to remove that excess oil buildup, allowing the hair strands to be lighter and more independent of each other. Additionally, the oilier hair gets, the more the hair shafts clump together and become matted and lifeless. Sea Salt Spray adds instant texture and volume to your hairĪs oil and product accumulate on the hair and scalp, hair shafts become heavy and weighed down. 
Let’s dive a little deeper into each of these. Sea Salt Spray can actually help remove toxins from your scalp and hair.Sea Salt Spray enhances the natural curl pattern of your hair.Sea Salt Spray adds instant texture and volume to your hair.What Does Sea Salt Spray Do?īeardbrand Sea Salt Spray does three things exceptionally well: 
More specifically, we’re going to be focusing on Beardbrand Sea Salt Spray. To answer that question, we’re doing a deep dive on sea salt spray to help you decide if it’s a styling product that will be a good fit for you. So wouldn’t salt suck the moisture right out of your mane? Naturally, the rise in popularity has also led to an assortment of questions - most notably - is it even a good idea to be putting salt in your hair? After all, when we think of salt, we typically think of it as a dehydrating mineral. Seems like sea salt spray is a win-win, right? Plus, bottled sea salt sprays aren’t full of the kelp beds, floating trash, and fish poop that make you want to wash your hair as soon as you get home. It’s enough to set your style, but unlike pomades or clays, sea salt spray leaves your hair soft and free to move.Īnd a bottled sea salt spray gives you and your flowing locks all the benefits of a day swimming in the ocean with a lot less of the drawbacks - crowds, sunburns, and sharks. Sea salt spray gives your hair just a touch of hold. And looking like you just spent the day hot-dogging in the surf with the light offshore winds blowing your hair into messy windswept perfection is equally as timeless (and cool). Hanging ten on a longboard in Malibu? That’s timeless cool. The same applies to your hair - long, flowing, and natural looks are where it’s at. Shirts have gotten bigger and boxier, denim has gone back to more relaxed cuts, and pleated trousers are no longer reserved for film noir and your Grandpa Joe. The biggest style trend to emerge in the 2020s is that everything is loose and flowing. Few hairstyling products have garnered as much attention over the past year as sea salt spray - and for good reason.
Back to Blog
Live traffic cameras el paso texas1/18/2024 
The current webcam image available on the external website. View from cameras on the border bridges of Stanton Bridge, Paso Del Norte Bridge and Zaragoza. Users can also unsubscribe at any time by logging in and unsubscribing their account. Ciudad Jurez - Several border crossings 16 km (9 mi) Tornillo - Railway crossing 50 km (31 mi) Las Cruces - Panoramic view 68 km (42 mi) Advertisements. Check Live Texas Highway Traffic Webcams/Cameras To Avoid Road Construction and Other Delays. Users can unsubscribe by clicking the “unsubscribe” link in email or text messages received. Subscribing to receive email and or text messages from the site is entirely voluntary. The email and phone information provided will not be provided to any other agencies or third parties for advertising or any other purpose. EL PASO, Texas Veterinarians and dog owners across the country have been concerned over a mysterious dog-respiratory illness that has reportedly been spreading across multiple states. This information is confidential and used strictly for the purpose of sending relevant information about New Mexico road conditions for road segments requested by the subscriber. To create an account and receive messages, the user must provide an email address and optionally a phone number and phone carrier to receive text messages. side and Government of México in the Mexican side.įor more information about FAST, SENTRI, and Global Entry visit the Trusted Traveler Programs section on the Information for Travelers web site users can subscribe to receive important information by email and or text message about road condition alerts and traffic information that occur on roads maintained by the New Mexico Department of Transportation. The Ysleta-Zaragoza bridge is owned by the City of El Paso in the U.S. The non-commercial bridge also has two pedestrian walkways to accommodate northbound and southbound pedestrian movements. The non-commercial bridge has two lanes for vehicles in each direction and one northbound SENTRI/Global Entry lane, also known as a Dedicated Commuter Lane ( DCL). 
The commercial bridge has two lanes for southbound traffic, one Standard lane for northbound traffic and one northbound FAST lane. Hundreds of migrants who are part of a recent influx are currently camping out outside the church. It was founded in 1893 and has been welcoming refugees from Mexico since the 1920s. This port of entry consists of two structures, one is an 804-foot, four-lane bridge for commercial traffic and the other is a five-lane bridge for non-commercial traffic. Sacred Heart Chuch is located in the heart of El Paso's Segundo Barrio. side and Waterfill Avenue on the Mexican side. Our Hosts Login El Paso, Texas Railcam Overview: Location: Tower 47 (Nearby) Traffic: Approximately 40 trains per day RRs: Union Pacific and Amtrak Time Zone: Mountain Standard Time (GMT -7) Membership Level:Engineer. There are no current restrictions for other pets such as rabbits, guinea pigs, ferrets and hamsters, so long as they appear to be in good health. A valid rabies vaccination is required for all dogs. It sees approximately 40 Union Pacific and Amtrak trains per day. will be examined at the border crossing for evidence of any diseases that could be transmitted to humans. The Ysleta-Zaragoza bridge is located in El Paso, Texas, at 791 S. The El Paso, Texas railcam is located near Tower 47.
Back to Blog
Halide precipitate tests1/17/2024 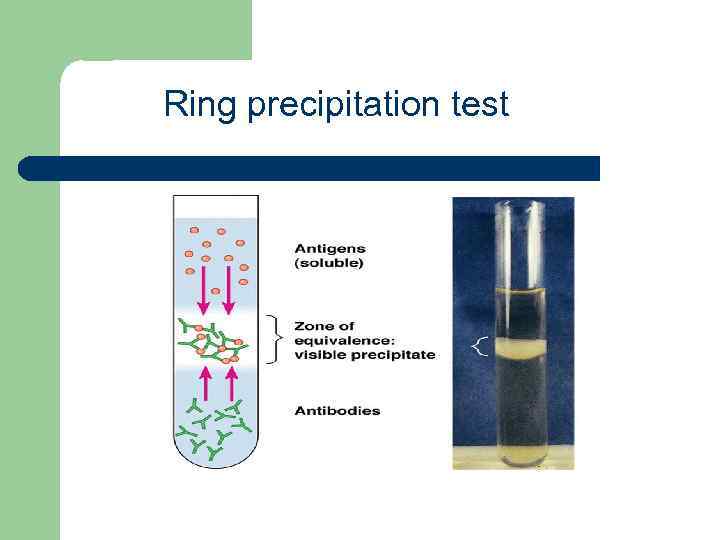
I only remember this as being the last test left over :c. The last test is testing for a sulfate, this is adding dilute hydrochloric acid and then barium chloride solution and if a white precipitate is formed then it's a sulfate. I would remember this by alphabetical order for both halide and precipitate colour, so Cream precipitate would be a Bromide, a White precipitate would be Chloride and a Yellow precipitate would be Iodide. Halide ions in solutions are detected using silver nitrate solutions. 
Their ions are called halide ions, eg chloride, Cl. 
The second test is testing for halides where you add dilute nitric acid and silver nitrate to the solution and the precipitate that forms is either white (for chloride), cream (bromide) and yellow (iodide). Chlorine, bromine and iodine are halogens. The way I remember this is that you are trying to find carbon dioxide and carbon is part of testing for a carbonate The first is testing for a carbonate you just add dilute hydrochloric acid and the gas that's formed you add into limewater, if it turns cloudy then carbon dioxide is produce so there is the presence of a carbonate. Transfer about 5 mL each of Mg(NO 3) 2, Ca(NO 3) 2, Sr(NO 3) 2, and Ba(NO 3) 2 solutions into four (4) labeled large test tubes.So there are 3 tests for the negative ions: Part 1: Reactions involving Group 2 c ations When you are finished with the experiment, pour the contents of the waste beaker into the inorganic waste container in the fume hood. Then place the splint in a roaring Bunsen. How to carry out a flame test Place a damp splint into some of the ionic compounds as a solid. test for halides - iodide precipitate colour. test for halides - bromide precipitate colour. While you are doing the experiment, collect your reaction mixture into a waste beaker. Add dilute nitric acid then silver nitrate, forms a coloured silver halide precipitate. Sodium fluoride solutions will react with glass and should not have long term contact with glass (short time periods with dilute solutions are not generally a problem). Contact may not be seen until after exposure to direct sunlight. Avoid direct contact with silver nitrate solutions. Silver nitrate (AgNO 3) solutions will react with skin and leave brown stains. The silver nitrate test is sensitive enough to detect fairly small concentrations of halide ions. There is no need to make this reaction go to completion. Aqueous silver nitrate was added and a cream precipitate was. The halogen atom is displaced as a halide ion: R X + OH R OH +X (1) (1) R X + O H R O H + X. Test for halide ions using HNO3/AgNO3(aq) An unknown sample was dissolved in water to make a colourless solution. silver iodide is yellow silver bromide is cream coloured. Each silver halide precipitate has a distinctive colour. Use of eye protection is recommended for all experimental procedures. Halide ions will then precipitate as silver halides. Section 2: Safety Precautions and Waste Disposal In this experiment, students will conduct a series of reactions involving some alkaline earth metal ions and halide ions. Students will determine observable trends and characteristic reactions within each group. Using the information gathered, students will determine the elements present in an unknown solution. Note that Be, Ra and At are not included in this experiment. We will be using the current designations of Group 2 (Be, Mg, Ca, Sr, Ba, Ra) and Group 17 (F, Cl, Br, I, At). e) Complete the following table to show the results of doing this. d) Write the ionic equation for the reaction involving iodide ions. Name the precipitate if you had iodide ions present. NOTE: Some older textbooks use prior group designations of Group 2A and Group 7A. You can test for the presence of the halide ions chloride, bromide and iodide by adding dilute nitric. Identify an unknown solution based on the properties observed for the ions. \)Ĭarry out reactions involving Group 2 cations and Group 17 halides.
Back to Blog
Epson wf 3520 windows 101/17/2024 
Also supports Box, Dropbox, Evernote®*1, Google Drive™ and Microsoft OneDrive™. 
Print photos, emails, webpages and files including Microsoft® Word, Excel®, PowerPoint® and PDF documents. Print, scan, and share directly from your iPhone®, iPad®, or iPod® Touch.
Back to Blog
C standard tuning1/17/2024 
Study note relationships rather than specific note clusters in chords and scales. This is just to get your bearings and is exactly the same process I teach students using standard E tuning.īeyond these root note reference points, study intervals rather than notes. With scales, after you've learned the notes across each string, you should be able to find the low root note positions for the most common scale patterns, and my lessons can be understood in exactly the same way, because I use interval labels rather than the notes themselves.įor example, a major scale pattern rooted at the 5th fret in C standard tuning would make it an F major scale because the low C string at the 5th fret is F.Ī harmonic minor pattern rooted at the 9th fret would be A harmonic minor because the low C string at the 9th fret is A. Open D major/minor becomes open A#/Bb major/minor Open E major/minor becomes open C major/minor With chords, start back at those open position chords.  
Starting with the open strings, you should be able to work this out and learn in the same time frame as E standard tuning. So I would start by learning the notes across the fretboard. The only thing new to learn is the notes across each string up the fretboard, as this will give you a reference point for, say, finding the root note of a given scale or chord. I didn't realize it might bite me in the (b)ass, to slow my development.įirstly, if you like the tuning, stick with it.Īs you're still in a form of standard tuning (so the same relative tuning as E A D G B e, albeit 2 whole steps down) the actual scale patterns and chord shapes/forms remain exactly the same. I know now that I should have learned it when I started playing Guitar 10 years ago but didn't, believing it would hinder my talent & staunch my creativity. Is there a simpler way to do it or would it be best for me to re-tune to standard & learn the standard scale & chords completely? I recently started studying the standard scale & trying to reverse translate it, but am having a hard time with it. I like this particular tuning & don't want to stop using it, as everything I play & do is set to it, helping me develop my tone. Now I find it difficult sometimes to locate scales & chords as quickly or easily as I should. Unfortunately, I did not take the time to learn the standard scales & chords before I changed tunings. The more you tune to that tuning, the faster and better the tuner will complete that tunings due to the learning algorithm.I tune my Guitar to C Standard (all strings down 2 whole notes) from low to high: Should you encounter any issues, tune in Single String Mode (push and HOLD Enter at selection of White A). Low C tuning is White bank A LED, Enter to activate Multi String Tuning, the tuner will perform the approach, now strum and let vibrate all strings for 3-5 sec., then mute all strings and pick the ones that are not in tune only. Now press UP or DWN consequently to toggle between the 6 banks (Red, Green, Blue / White, Yellow and Magenta) Press ENTER.ģ.Use LEFT/RIGHT button to activate low tunings and additional preset banks ( e B G LEDs flash GREEN ) or to deactivate low tunings and addition-al preset banks ( D A E LEDs flash RED ).Ĥ.Press ENTER = low tuning/additional banks settings stored.ĥ.Press and hold ON to switch off the tuner.Ħ.Press ON two times for tuning selection. Press the LEFT/RIGHT button to select the YELLOW G LED. Press the LEFT/RIGHT button to select YELLOW D. However, you can also convert any of these tuning preset banks to store presets.ġ.When the GForce is switched off, press ON three times. The MAGENTA bank and the BLUE bank, defaults to be write-enabled for storing user tuning presets. The WHITE and YELLOW banks default to write-protected presets for low tunings (heavier gauge strings are recommended). This function activates three additional tuning preset banks. Unblocks three additional tuning preset banks 
You have to activate the low tunings banks:ĪCTIVATE LOW TUNINGS AND/OR ADDI-TIONAL USER TUNING PRESET BANKS Looking at the instructions there is no "press this 3 times, press this 2 times" style directions for C standard. I've installed thicker strings to have one lower tuned guitar.
Back to Blog
9mm winchester white box1/17/2024 
In this case, Winchester has supplied us with a little data to work off of, noting 5 yard Velocities of 1190 fps and energies of 353 ft-lbs out of the barrel. I used this data to position myself at the correct distance of 15 feet form the Chronograph for testing and marked my position so that I could return to it after each reloading for each test group. To the left I have included the label from the Winchester 9mm White Box Ammunition Factory Packaging. Many manufacturers printed data on the box, some do not. I have recorded the outcome of these measurements below as well as photographs of each cartridge and its components throughout the disassembling process. The individual 9mm cartridges are then disassembled, and set aside to be measured and weighed. The 5 samples from the 9mm Winchester White Box ammunition were first weighed and measured in their factory loaded state to record overall variances. More details about my process and the equipment used for testing the Winchester 9mm White Box Ammo are at the bottom of this page. To test the Winchester White Box 9mm ammo I first select 5 samples from the 50 round box, disassemble the components and take measurements in terms of both physical sizes as well as weight in grains. In the case of the Winchester 9mm White Box Target ammo, i found this to be particularly true. For the most part, this generally seems to be true. The general consensus is that factory line assemblies do not produce the same quality or employ the same close tolerances as ammunition that is loaded by hand. Many of the large ammunition manufacturers produce their low end target ammo using automated machines, causing debate over exactly what the tolerances really hold up to and how much variance there is from one cartridge to the next. There has been much discussion in the past regarding factory loaded ammunition and quality control issues. Winchester White Box 115 grain 9mm Cartridges 
The Winchester White Box 9mm ammo falls into the lower end of that spectrum and serves as a mostly economical solution for those that would like to practice their handgun skills more frequently on a budget, but would still like the reassurance of a brand name American company manufacturing their ammunition. In their lineup of products, Winchester offers products from the expensive and well made down to the cheap and highly economical. Winchester is an old American company and with that comes lots of history, a wide selection of firearms spanning centuries, and a good selection of ammunition, reloading components, and firearms accessories. The 115 grain variation of this 9mm ammunition, which is what we are testing in this article, can be found at most retailers where firearms are sold and can be had anywhere from $10-22 USD depending on where it is purchased. The Winchester Target 9mm Ammo, also known as Winchester White Box 9mm ammo, is a popular and fairly economical choice for target practice in 9mm Parabellum. About the Winchester White Box 9mm Ammunition
Back to Blog
Pickering emulsion disc particles1/17/2024 
Modify-then-exfoliate is more efficient than the mask-and-modify method as it allows bulk reactions rather that limiting reactions to a two-dimensional interface. Masking can be achieved on either emulsion interfaces 24 or solid substrates 25, but regardless of procedure, reaction can be carried out only at the interface, rendering these methods tedious and not suitable for practical use. Mask-and-modify is a very common strategy for Janus platelet synthesis 23. Although the amphiphilic platelets have the potential to be used to stabilize emulsion, the current synthesis methods for such platelets is not reliable. Such amphiphilic structure of a nanoplate is also known as a Janus platelet, which has been previously reported by our group 21, 22. To minimize interfacial tension, asymmetric surface modification may be applied to satisfy different wetting preferences on corresponding sides of the platelet. Interfacial energy, in turn, is related not only to particle geometry, but is also controlled by surface wetting property. These merits contribute to the enhanced emulsion stability.Įmulsion stability varies directly with interfacial energy, a characteristic which has been well analyzed in other peer works 19, 20. Lastly, the two-dimensional configuration has higher efficiency in terms of material utilization compared to spherical or cylindrical geometries 18. Also, the large surface coverage area significantly increases the magnitude of energy required to remove platelets from the interface. 
There are several merits for doing so: firstly, the two-dimensional sheets can provide greater surface coverage than other configurations, which can prevent the emulsified phase from diffusing to the continuous phase. Among different particle morphologies, the idea of applying two-dimensional disks or platelets to stabilize emulsion attracts more attention 15, 16, 17. In related research, a variety of nanoparticles with different morphologies have been used to stabilize Pickering emulsions, such as zero-dimensional nanoparticles 12, one-dimensional nanowires 13, and one-dimensional nanotubes 14. Compared to emulsions stabilized by conventional molecular surfactants, Pickering emulsions are more stable due to their resistance to coalescence and Oswald ripening 3, 4, 5, which makes them perfect candidates for industrial application in foods 6, 7, mining 8, 9, pharmaceuticals 10 and cosmetics 11. The structures of Pickering emulsions and foams are stabilized with solid particles instead of molecular surfactants. Particle-stabilized emulsion, also known as Pickering emulsion, might provide a solution 2. Conventional EOR is carried out with molecular surfactants however, harsh formation conditions, such as high temperature or high salinity, can decrease the efficiency of molecular surfactants or polymer systems. Enhanced oil recovery (EOR) is a common well stimulation method to increase productivity of oil wells. Unless certain stimulation measures are taken, production on an aged well will be less economically viable for oil recovery. On the production side, production from aging wells will be reduced from current levels. Energy Information Administration’s (EIA) projection, fossil fuels will supply 75 quadrillion (10 15) BTU of energy by 2050 to meet the world energy demand 1. Similar content being viewed by othersĭespite the development of renewable energy sources, fossil fuel will remain as the dominant energy resource in the global energy supply for decades. This research provides important insights for the design and synthesis of two-dimensional Janus colloidal surfactants, which could be utilized in biomedical, food and mining industries, especially for circumstances where high salinity and high temperature are involved. Furthermore, we demonstrate the application of JNPS for enhanced oil recovery with a microfluidic flooding test, showing a dramatic increase of oil recovery ratio. The phase behaviors of water/oil emulsion generated by these novel platelet surfactants were also investigated. The microstructural characterization of solidified polystyrene emulsions indicates that the emulsion interface is evenly covered by JNPS. Such colloidal surfactants are found to be able to stabilize Pickering emulsions of different oil/water systems. Here we report a straightforward and cost-efficient strategy to develop Janus nanoplate surfactants (JNPS) from an aluminosilicate nanoclay, halloysite, by stepwise surface modification, including an innovative selective surface modification step. Combining the advantages of molecular surfactants and particle-stabilized Pickering emulsions, Janus colloidal surfactants generate remarkably stable emulsions. Janus colloidal surfactants with opposing wettabilities are receiving attention for their practical application in industry. |
 RSS Feed
RSS Feed


